Hanta Virus (Andes strain) How it Works
Why This Virus became a problem
How does the Virus Infect?
How does a virus that mostly infects mice become a virus that infects humans? What change happens to allow that and how does the current Hanta virus (Andes strain) compare to other versions? Today we will answer that question with our latest updated information from the genes that have been analyzed.
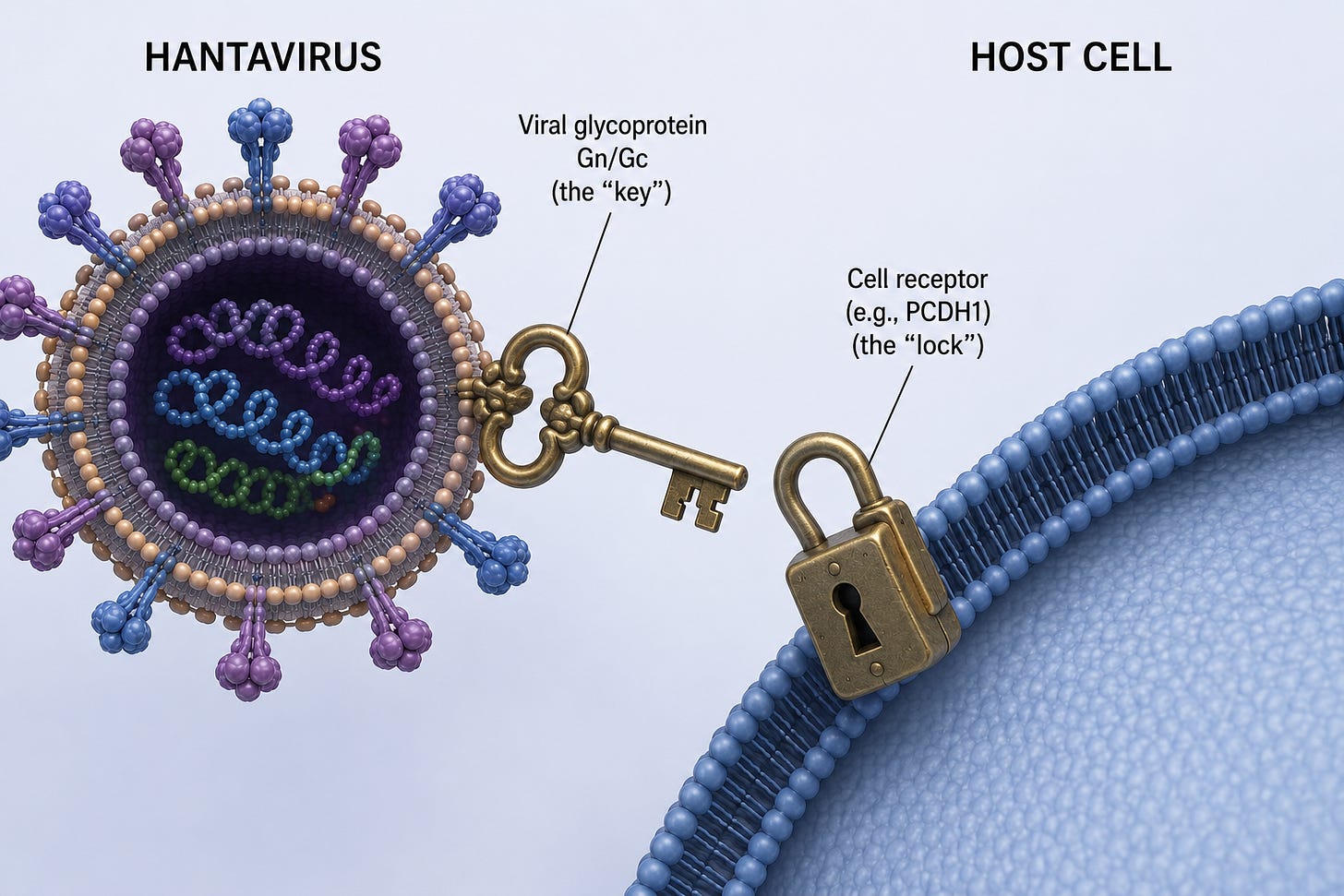
Think Lock and Key
A virus doesn’t just enter a cell and begin replication. A virus must have the right key to unlock the door to enter that cell. Not every cell of the human body is subject to infection from a virus, because not every cell has the right lock that can be opened by the viral key.
One of the genes code the Hanta virus “key” is on the protein surface membrane of the virus, and is coded by one of the genes in the Hanta virus. The only cells in the human with this lock are the cells of the endothelium, or lining of the blood vessels. Most of these are in the lungs, some in the kidneys.
The M protein of the Hanta Virus determines if it can be transmitted by mice or people. So the M protein determines if the virus can be transmitted from human to human, and how well it can enter the blood vessel wall.
Mutations in Viruses
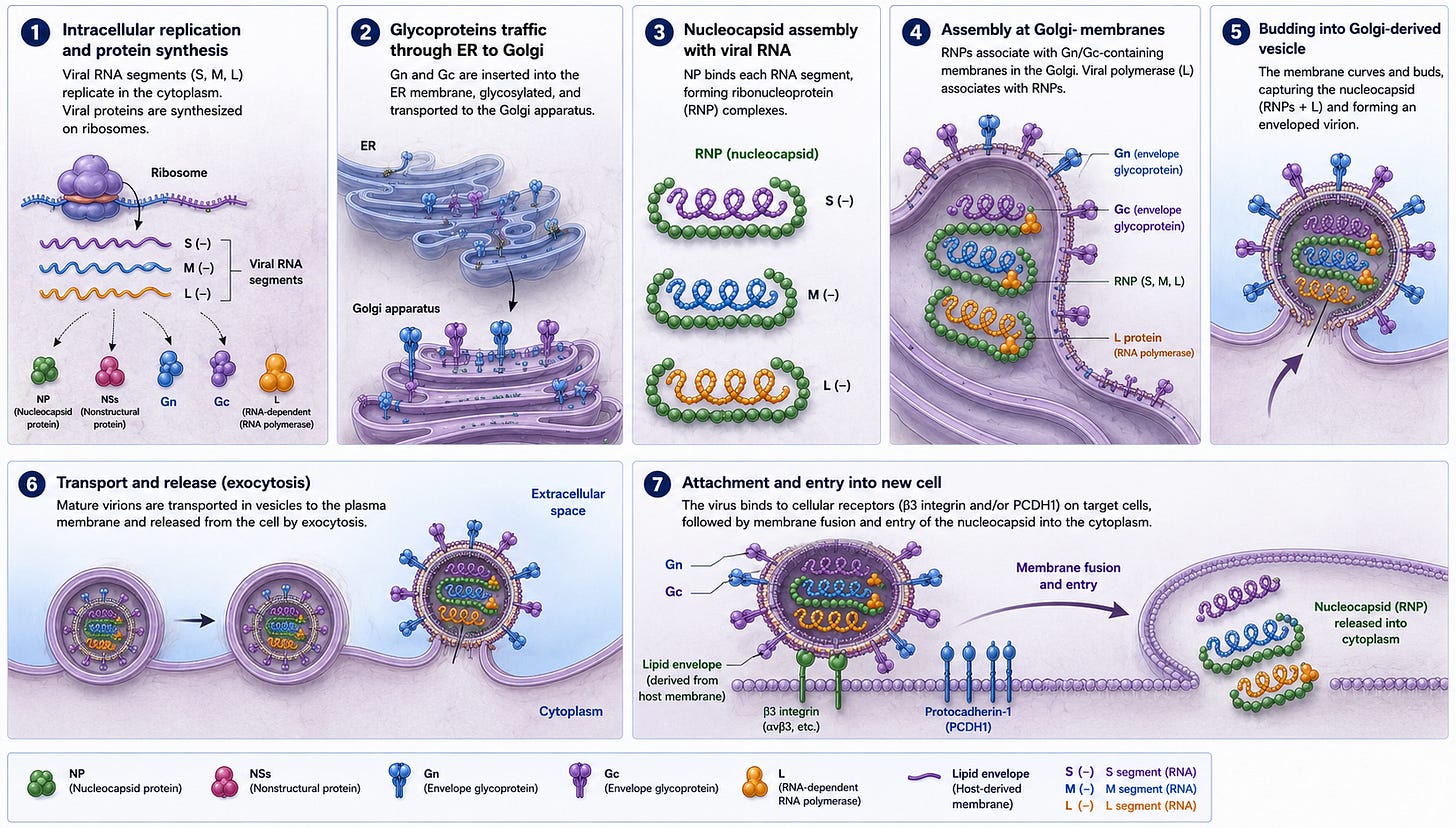
Mutations in viruses happen all the time. And with RNA viruses, these mutations happen more than in DNA viruses. These mutations happen when the virus replicates, which is after it enters the cell.
When Hanta virus enters the endothelial cell, it begins to replicate, and it makes hundreds of copies of its proteins, and then they begin to reassemble into a virus.
If a mutation occurs and the virus proteins don’t fit together, that virus isn’t viable. Most of the time, over 99 percent of the time, these or other insignificant mutations occur.
Take Measles virus. It is an RNA virus that replicates in immune cells of the lungs (macrophages, lymphocytes, dendritic cells). But measles has a very specific manner in which it must be put together. Think an Ikea project.
But if those cell proteins with mutations don’t fit, then that measles virus doesn’t get put together and isn’t infective or effective.
Measles as an Example of Mutations
Measles is actually a great example of why not all RNA viruses mutate equally.
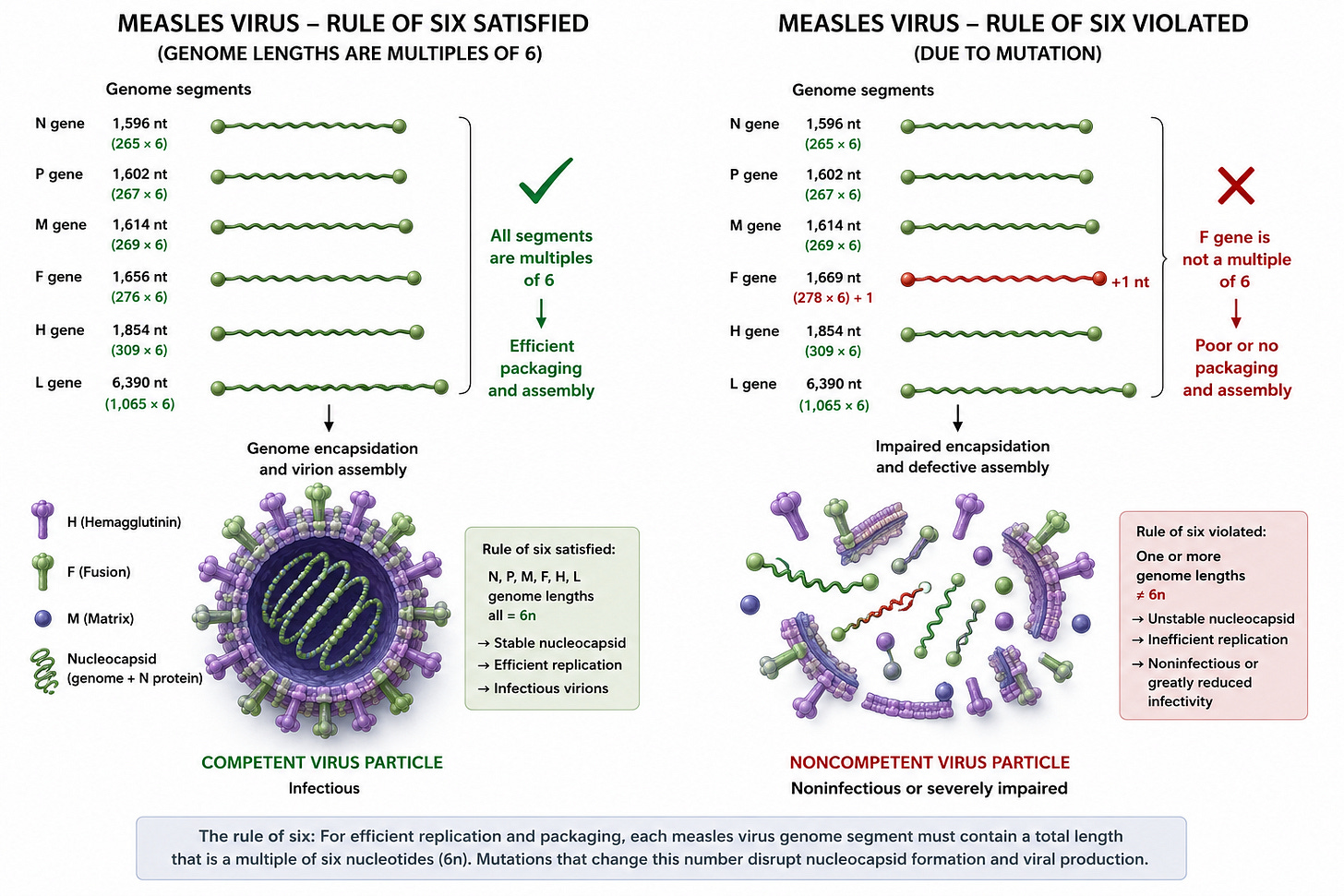
The measles virus follows something called the “rule of six.” Its genome works best when the total RNA length is divisible by six nucleotides, because the nucleocapsid proteins wrap the RNA in units of six. If mutations disrupt that geometry, the virus often cannot assemble or replicate efficiently.
So yes — measles mutates. All RNA viruses do. But most mutations are evolutionary dead ends, because they break the precise structural fit required for replication and packaging.
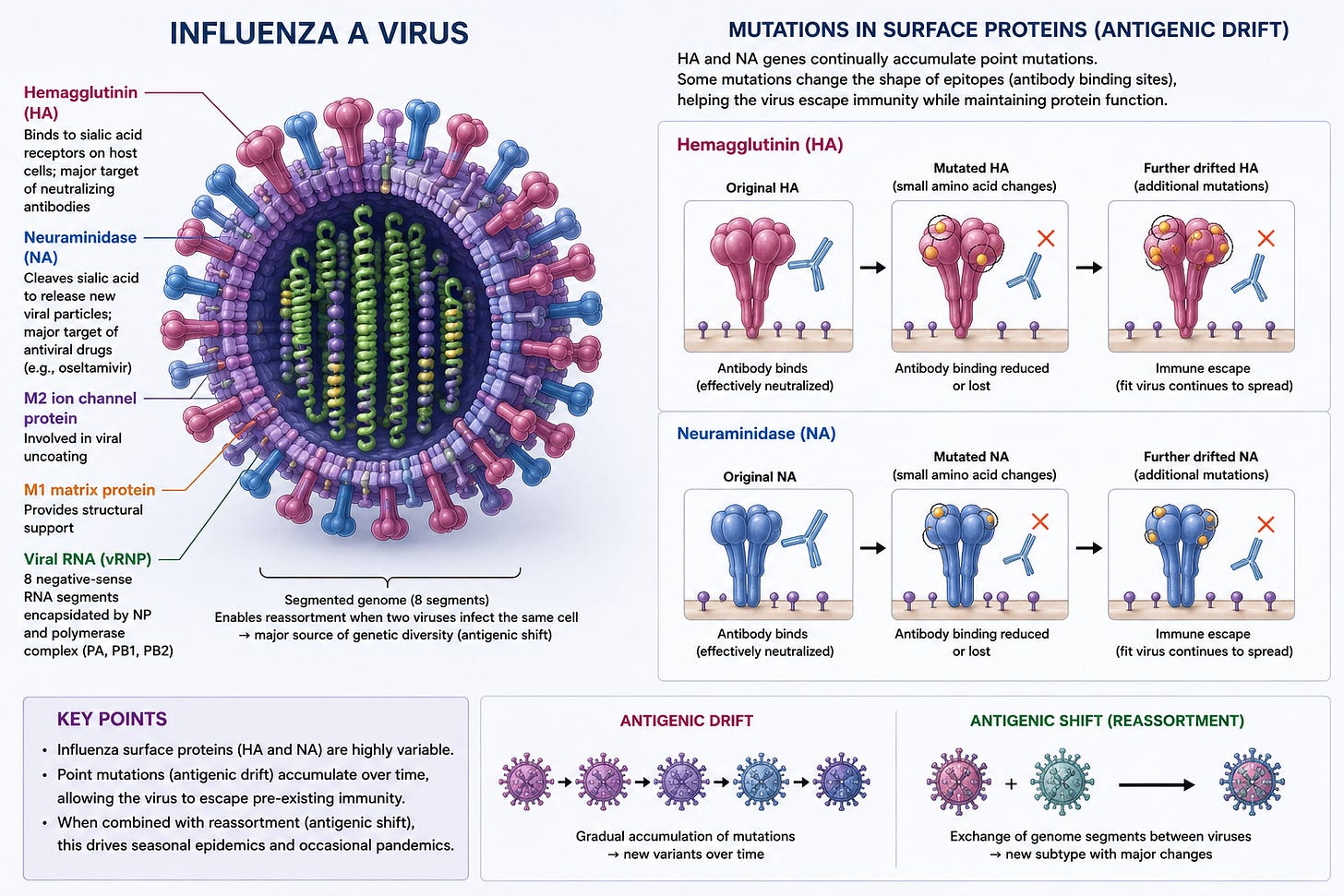
Influenza is very different
Influenza tolerates mutations much more easily in its surface proteins — hemagglutinin and neuraminidase. Even small changes can help it evade immunity while still remaining functional. On top of that, influenza has a segmented genome, so two strains can swap segments entirely (“reassortment”), creating major antigenic shifts.
This is why influenza changes quickly - every year or more. And why we must make vaccines annually for our boosters. Some years, those mutations can cause the virus to be much more contagious, and some years the virus can cause much more illness (hospitalizations, ICU admissions and deaths). Think Spanish Flu, Hong Kong flu, etc as years when we had rough times.
Hantaviruses sit somewhere in between.
Like measles, hantaviruses appear under fairly strong purifying selection. Their glycoproteins, nucleocapsid proteins, and polymerase are structurally constrained, and many mutations reduce fitness. But unlike measles, hantaviruses do not follow the strict “rule of six.”
So:
Measles = highly constrained assembly system
Influenza = mutation-tolerant shape-shifter
Hantavirus = constrained, but still capable of important adaptive mutations
That is why the measles vaccine remains effective decade after decade, while influenza vaccines must constantly change.
We have mapped the current Hanta Virus
We have the genome of the current Hanta virus, from those patients the virus infected, including two who died, and they are the same virus.
This gene sequencing has told us that the mutations are new, and all in the M protein responsible for that viral key. They do not tell us if this virus is more contagious, but we do know that this virus kills about 38% of the people it has infected. So this is a dangerous variant. It is similar to the previous Andes versions found in Patagonia (where the couple was bird watching).
The genetic sequence cannot tell us if this virus will cause a major issue.
Chances of a pandemic are low, but it is a warning
The world is getting smaller.
More people are traveling into remote regions once biologically isolated — Patagonia, the Congo, Arctic regions, deep forests, expanding agricultural zones. That means humans are increasingly encountering viruses that have been quietly evolving in animals for centuries.
Most of those spillover events go nowhere. The virus cannot enter human cells efficiently. Or it replicates poorly. Or it burns out quickly.
But occasionally, evolution finds a workable combination.
That is why the question is not whether humanity will face another pandemic someday. The question is when.
I do not believe the current Andes hantavirus outbreak will become a global pandemic. Right now, the evidence does not support that conclusion.
But it is a warning.
A warning that zoonotic spillover events are increasing.
A warning that viral surveillance matters.
A warning that vaccine platforms, sequencing capability, and public health infrastructure are not luxuries.
It is also a reminder that withdrawing from global public health organizations like the WHO was a poor decision, and weakening institutions like the CDC by firing experienced personnel was equally shortsighted. Viruses do not respect borders, political parties, or national ideology. Surveillance systems work best when scientists, laboratories, and public health agencies share information rapidly across countries.
Pandemic preparedness is not global charity.
It is national self-defense.
Nature is constantly running evolutionary experiments.
Most fail quietly.
We only remember the ones that succeed.
Dr. Simpson began his career doing viral DNA anatomy and described how this could be used to identify new strains of virus and follow others in epidemics. He decided he liked people better than Petri dishes and became a surgeon instead of continuing as a research scientist. Who knew forty years later, that those studies would become important to the modern world.
Buchman TG, Simpson T, Nosal C, Roizman B, Nahmias AJ. The structure of herpes simplex virus DNA and its application to molecular epidemiology. Ann N Y Acad Sci. 1980;354:279-90. doi: 10.1111/j.1749-6632.1980.tb27972.x. PMID: 6261647.